Vanadium flow batteries are an interesting project for DIY hackers using readily available materials.to that effect [Cayrex2] YouTube offers a look at a small, self-contained flow battery made with off-the-shelf parts and some 3D printing. The video (embedded below) is part 5 of the series, detailing the final construction, charging and discharging process. His first four parts in the series are Part 1, Part 2, Part 3, and Part 4.
The concept of flow batteries is as follows. Flow batteries store energy through chemical changes, rather than storing energy as chemical changes on the cell's electrodes or as local chemical changes in an electrolyte layer.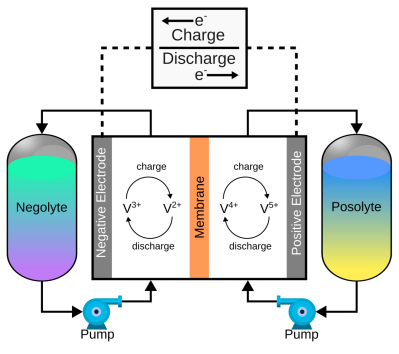 of a pair of electrolytes. These are kept outside the cell and connected to a pair of pumps. The capacity of a flow battery depends on the volume and concentration of the electrolyte, not the electrodes. This means that for a stationary installation, you will need a pair of larger tanks to increase storage capacity. A 4 MWh containerized flow battery has been installed in various locations where renewable energy storage requires a buffer to smooth the flow of power. The great thing about vanadium flow batteries is the versatility of vanadium itself. Because it can exist in four stable oxidation states, flow batteries can utilize it on both sides of the reaction cell.
of a pair of electrolytes. These are kept outside the cell and connected to a pair of pumps. The capacity of a flow battery depends on the volume and concentration of the electrolyte, not the electrodes. This means that for a stationary installation, you will need a pair of larger tanks to increase storage capacity. A 4 MWh containerized flow battery has been installed in various locations where renewable energy storage requires a buffer to smooth the flow of power. The great thing about vanadium flow batteries is the versatility of vanadium itself. Because it can exist in four stable oxidation states, flow batteries can utilize it on both sides of the reaction cell.
In this build, the reaction plate at the heart of the cell is printed with an “ABS-like” resin. They consist of a flat plate with a through hole for the clamp, a central opening housing the charge collector electrode, a protective layer, and an ion exchange membrane. Pipes at the ends provide small holes at the inner ends of each end of the flow region to allow electrolyte to pass to and from the external reservoir. Resin printing was chosen for its strength and, most importantly, its smooth surface, which helps prevent leakage. The electrode is a copper sheet with a protective layer of conductive HDPE and a second layer of graphite felt. The last layer conducts electrons to the HDPE and copper electrodes while allowing lateral electrolyte flow from the reservoir. A central Nafion-based ion exchange membrane prevents mixing of positive and negative electrolytes. However, an initial test using baking paper also works for a while. Each half of the battery is filled with vanadium pentoxide and sulfuric acid.
Although the theoretical cell voltage is centered around 1.5 volts, sufficient current must be available even with such a small electrode area. It was fun to watch his two halves of the cell visually change their electrolyte status during the charging process. Vanadium produces stunning colors in various oxidation states. If you want to play along at home, you can find his STL files for the 3D printed parts on his Cayrex2 Patreon site.


