Two US researchers have developed a simple, cheap and effective method to recycle catalytic organic iridium waste into a synthetically important iridium precursor. This is the first laboratory-scale example of such a recycling process and could be a practical and cost-effective option for many academic research groups working with this rare element.
“Precious metal waste from academic laboratories was previously collected separately and sent back to suppliers. Suppliers recycle these concentrated waste streams and repay us as new starting material. '', explains Ulrich Hintermeer, a catalytic researcher at the University of Bath in the UK. “These recycling programs were discontinued about 10 years ago. They were too expensive to run safely, so now all these precious elements are treated like other laboratory waste. and then incinerated in large-scale facilities.'' From a sustainability perspective, that's crazy. ”
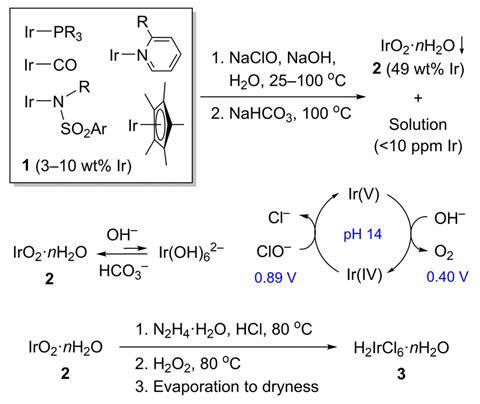
Now, Travis Williams and Valery Cherepakhin of the University of Southern California have proposed an alternative method for processing organic iridium waste. Their method uses sodium hypochlorite (bleach) and other inexpensive reagents such as sodium hydroxide and sodium bicarbonate to produce hexachloroiridic acid. “For other precious metals, [recycling industries] I'm trying to go to ore…I wanted to go to chloridate. [starting point] All commercial iridium complexes are made from it,” Williams says.

The process begins by oxidizing organic iridium waste in a sodium hydroxide solution, followed by brief heating with bleach to produce an iridium hydroxide intermediate. This is precipitated from solution using excess sodium bicarbonate at 100 °C before redissolving in hydrochloric acid in the presence of hydrazine. Adding hydrogen peroxide to the solution oxidizes the remaining Ir(III) and destroys the excess hydrazine, after which the solution evaporates to yield hexachloroiridic acid, which can be used without further purification. argue Williams and Cherepakhin. To demonstrate this, they used the resulting hexachloroiridic acid as a starting material for their synthesis. [(1,5-cyclooctadiene)IrCl]2 The yield was 87%.
Catalyst experts are excited about the idea. “With the price of precious metals rising, this is a huge incentive for researchers, especially if they can recover iridium in an easy and safe way. We expect there will be a lot of interest in this research… would like to try the procedure in their own lab,” commented Loi Do from the University of Houston, USA. And Andreas Falz of the University of Basel in Switzerland says, “For research groups that use iridium and obtain large quantities of iridium waste, this provides a good solution.” This looks like a very good solution to a niche problem and they solved it well. ”


