
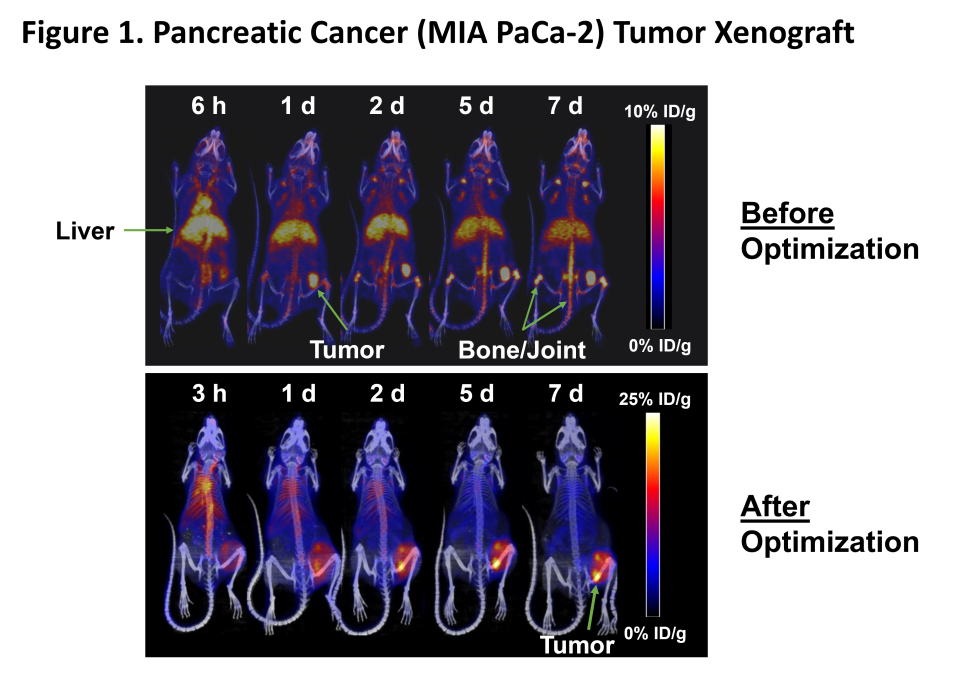
Figure 1
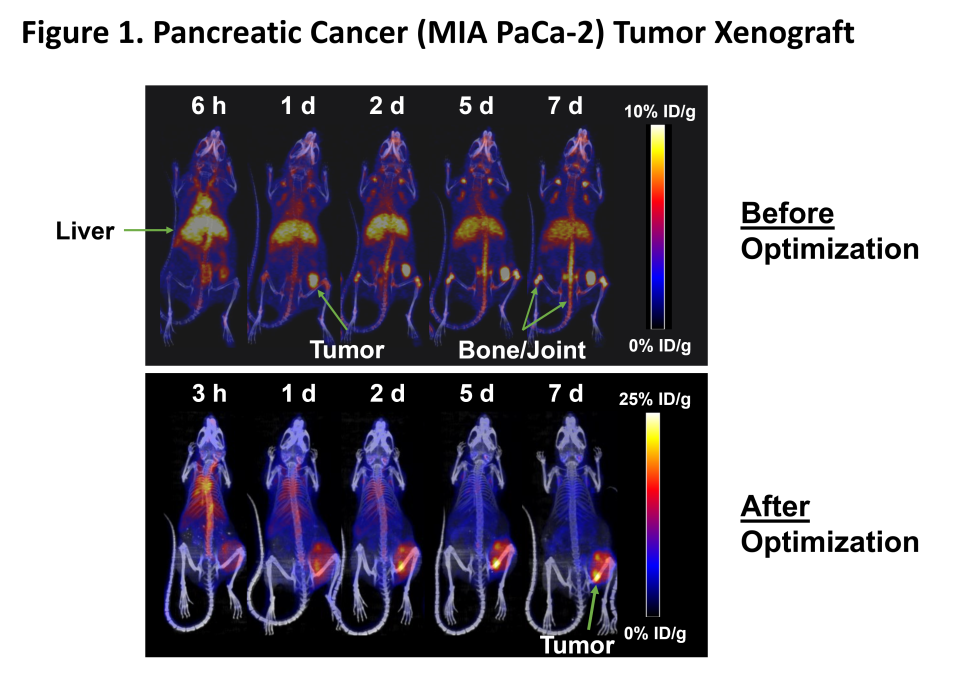

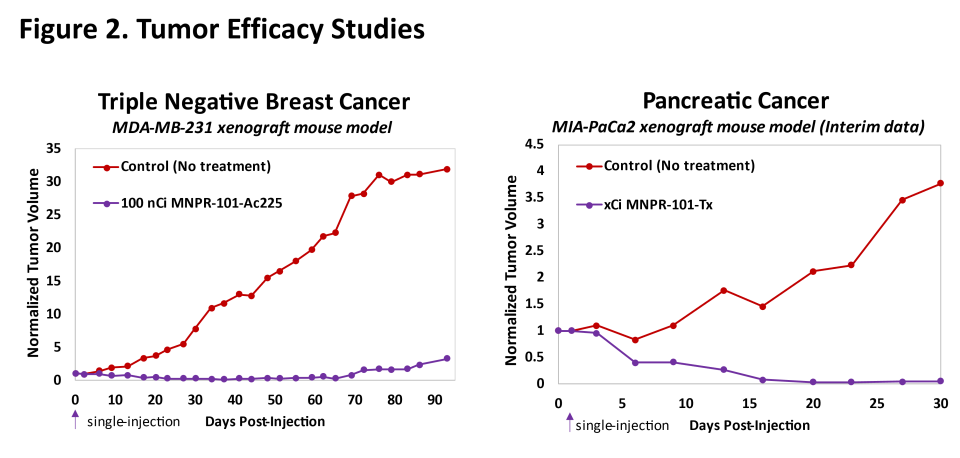
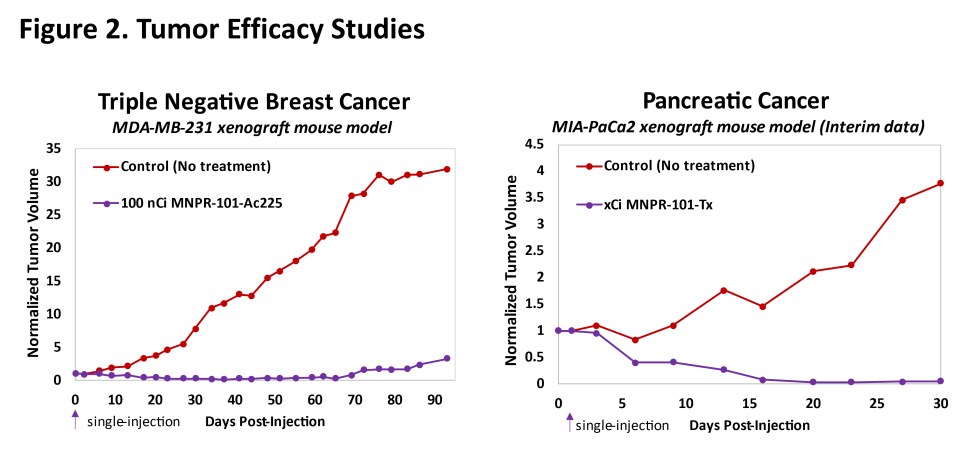
Figure 2.

WILMETTE, Ill., Feb. 22, 2024 (Globe Newswire) — Monopar Therapeutics Inc. (Nasdaq: MNPR) is a clinical-stage biopharmaceutical company focused on developing innovative treatments for cancer patients. Today, we announced promising preclinical imaging and treatment efficacy data. For the MNPR-101 radiopharmaceutical program. This new first-in-class radiopharmaceutical program targets cancers that express the urokinase plasminogen activator receptor (uPAR), including triple-negative breast, colorectal, and pancreatic cancers. Contains most of the.
MNPR-101 Imaging Radioisotope Binding
Maximizing the dose delivered to the tumor compared to normal tissue is of paramount importance in radiopharmaceutical treatment. Figure 1 below shows before and after optimization of Monopar's radiopharmaceutical contrast agent, MNPR-101-Zr, for advanced solid tumors that express uPAR. As shown in this preclinical positron emission tomography (PET) continuous image time series, Monopar's in-house radiopharmaceutical development team has shown that MNPR-101 can be used in tumors while minimizing uptake in healthy tissue. -We were able to significantly increase Zr uptake. High specificity and sustained tumor uptake are evident in the following optimized panel.

MNPR-101 Combined with therapeutic radioisotopes
Preclinical data to date demonstrate convincing and sustained antitumor efficacy with MNPR-101 combined with therapeutic radioisotopes. Figure 2 below shows preclinical efficacy data in triple-negative breast and pancreatic cancer human tumor xenograft mouse models utilizing two different therapeutic radioisotopes conjugated to MNPR-101. One of these radioactive isotopes has already been identified as actinium-225 (Ac-225). Both results show that the tumor was almost completely eliminated after a single injection of the radiopharmaceutical. These studies demonstrate the potential for MNPR-101-based radiopharmaceuticals to provide highly meaningful clinical benefits to patients.

Monopar recently announced that it has received Human Research Ethics Committee (HREC) approval to begin a Phase 1 dosimetric clinical trial of MNPR-101-Zr in patients with advanced cancer in Australia. “As we prepare to begin this clinical trial, we are encouraged by the significant, precise and sustained accumulation seen within tumors and the corresponding therapeutic efficacy in preclinical human tumor xenograft models.” Chief Operating Officer Andrew Sittadine said. “We aim to present these promising preclinical results at an upcoming scientific conference.”
About Monopar Therapeutics Co., Ltd.
Monopar Therapeutics is a clinical-stage biopharmaceutical company focused on developing innovative treatments for cancer patients. Monopar's pipeline consists of camsirubicin in Phase 1b for the treatment of advanced soft tissue sarcomas. MNPR-101 in Phase 1 for radiopharmaceutical use in advanced cancer. and an early-stage camsylubicin analog, MNPR-202. For more information, please visit www.monopartx.com.
Forward-looking statements
Statements contained in this press release regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. “may”, “would”, “might”, “should”, “expect”, “plan”, “anticipate”, “intend”, “believe” Words such as “estimate,” “anticipate,” “anticipate,” “may,” “continue,” “target” and similar expressions are intended to identify forward-looking statements; Not all forward-looking statements contain these specific words. Examples of these forward-looking statements include: These studies demonstrate the potential for MNPR-101-based radiopharmaceuticals to provide highly meaningful clinical benefits to patients. And we said we aim to present these promising preclinical results at an upcoming scientific meeting. Forward-looking statements involve risks and uncertainties including, but not limited to: that future preclinical or clinical data may not be as promising as historical data; Phase 1 clinical trials have not been initiated and enrolled. MNPR-101-Zr may cause unexpected serious side effects or may not be imaged or effective against human cancer tumors. the possibility that HREC may place the Phase 1 trial on clinical hold at any time; and the significant general risks and uncertainties surrounding the research, development, regulatory approval and commercialization of contrast agents and therapeutic agents. Actual results may differ materially from those expressed or implied by such forward-looking statements. The risks are detailed in Monopar's filings with the Securities and Exchange Commission. All forward-looking statements contained in this press release speak only as of the date on which they are made. Monopar undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date such statements are made. The forward-looking statements contained in this press release represent Monopar's views only as of the date of this press release and should not be relied upon as representing Monopar's views as of any subsequent date.
contact:
Monopar Therapeutics Co., Ltd.
PR for investors
Kim R. Tsuchimoto
CFO
kimtsu@monopartx.com
Follow Monopar on social media for the latest information.
Twitter: @MonoparTx LinkedIn: Monopar Therapeutics
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/3a5deb93-2122-4ee4-9ecb-745d9cbf923b
https://www.globenewswire.com/NewsRoom/AttachmentNg/cff0bf6c-68e7-440d-b721-5a5d77c28967




