

Twelve weeks of treatment with bexotegrast 160 mg reduced total collagen in the lungs
Compared to increase with placebo, measured by PET imaging
Improvement in FVC and reduction in cough severity reported in patients treated with bexotegrast
Compared to placebo at all time points
Bexotegrast 160 mg was well tolerated over 12 weeks of treatment
No serious adverse events or discontinuations
SOUTH SAN FRANCISCO, Calif., May 14, 2024 (Globe Newswire) — Pliant Therapeutics, Inc. (NASDAQ: PLRX) today announced topline data from a 12-week, randomized, double-blind, placebo-controlled trial of bexotegrast. announced. (PLN-74809) was conducted at Massachusetts General Hospital to assess changes in total collagen levels in the lungs of patients with idiopathic pulmonary fibrosis (IPF). IPF is a disease characterized by excessive collagen deposition in the lungs.
Patients treated with bexotegrast showed a decrease in total lung collagen after treatment as measured by positron emission tomography (PET) imaging, compared with an increase in total lung collagen in the placebo group, and a reduction in fibrosis resolution. suggested the possibility. Patients treated with bexotegrast showed improved forced vital capacity (FVC) and reduced cough severity at all time points compared to placebo. Bexotegrast 160 mg was well tolerated over 12 weeks with no drug-related serious adverse events (SAEs) or discontinuations.
The study evaluated bexotegrast at 160 mg once daily compared to placebo and measured changes in total lung collagen after 12 weeks of treatment in 10 patients with IPF. Patients underwent PET imaging with collagen-bound radiotracer at baseline and week 12.
The study enrolled seven patients in the active group and three in the placebo group. Eight out of 10 enrolled patients were receiving standard treatment, with the majority receiving nintedanib.
Patients treated with bexotegrast 160 mg showed decreased total collagen levels in the lungs after 12 weeks of treatment, suggesting possible reversal of fibrosis
The primary endpoint of this study was assessment of change in standardized uptake value (SUV). 68GA-CBP8, a PET ligand that binds to type 1 collagen. Type 1 collagen is the main type of collagen produced in the lungs as a result of IPF.1 An increase in SUV in the lungs indicates an increase in total collagen in the lungs and potential progression of the disease. IPF patients have been shown to exhibit higher SUV values compared to healthy subjects.2 Additionally, patients with increased lung total collagen on PET imaging had an increased risk of death.3
After 12 weeks of treatment, bexotegrast-treated patients showed a decrease in SUV in the lungs compared to the increase seen with placebo. This decrease in SUV indicates a decrease in total collagen in the lungs in the treatment group and suggests a possible reversal of fibrosis.
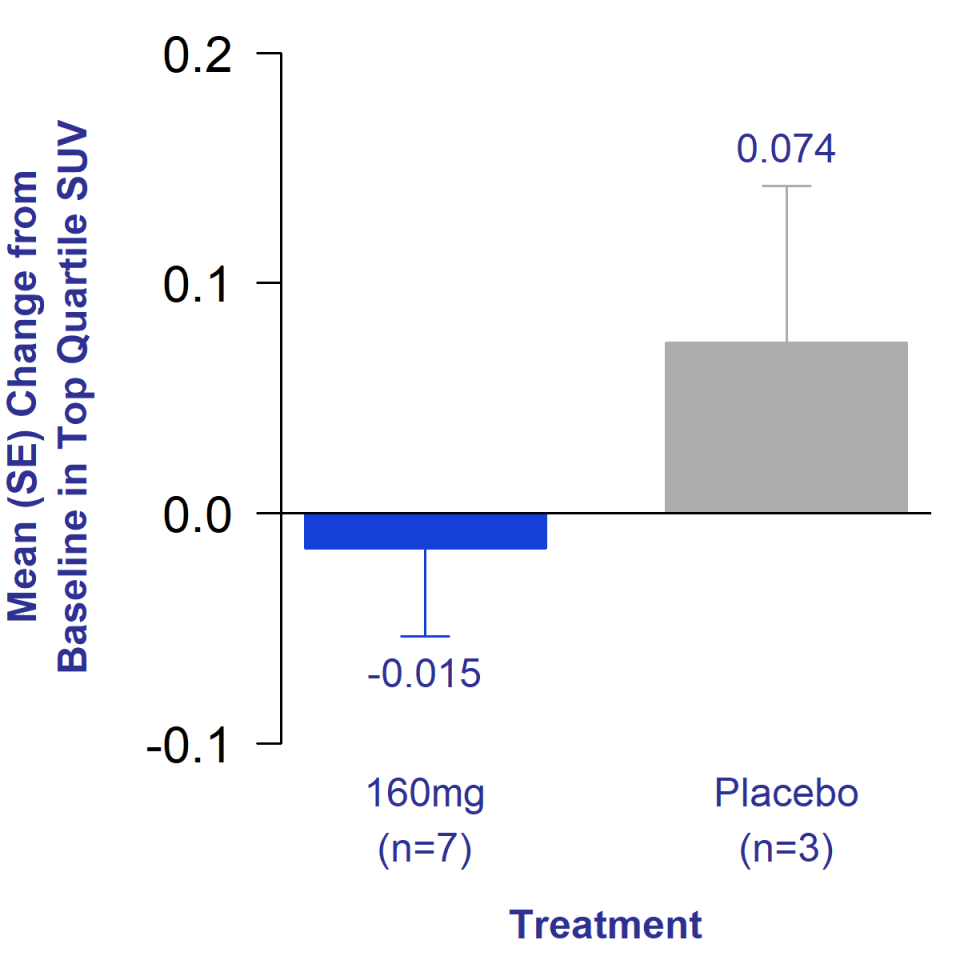

Figure 1. Mean change from baseline in collagen PET tracer uptake after 12 weeks
Patients treated with bexotegrast showed improvement in FVC, cough severity, and fibrosis biomarkers at all time points
The study's exploratory efficacy endpoints evaluated changes in FVC, forced vital capacity percent predicted (FVCpp), patient-reported cough severity, and fibrosis biomarkers. Patients treated with bexotegrast experienced improvements in lung function as measured by FVC and FVCpp, clearly distinguishable from placebo at all time points.
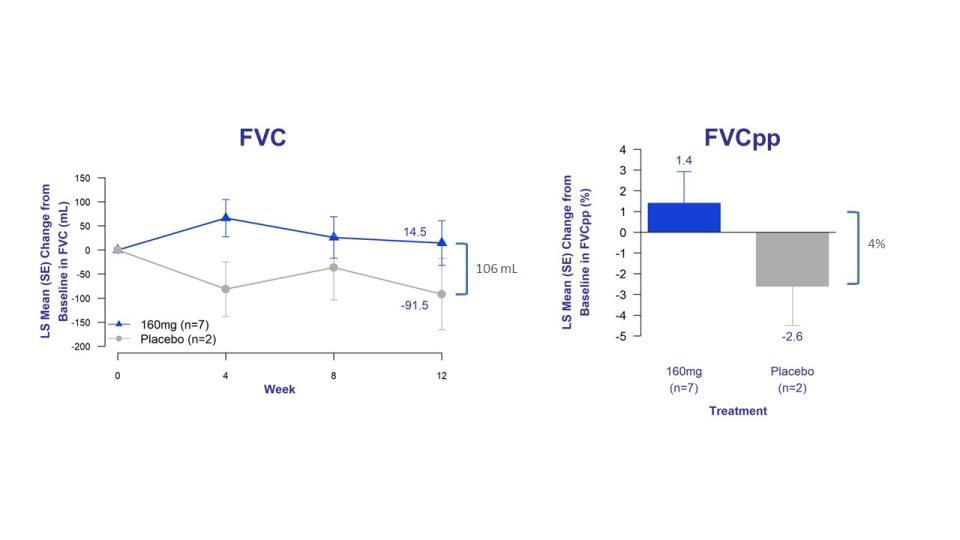

Figure 2. Changes in FVC and FVCpp from baseline on bexotegrast 160 mg over 12 weeks
Chronic cough in IPF is often intractable and debilitating.Four It is an independent predictor of disease progression and may predict time to death or lung transplantation.Five At all time points, bexotegrast-treated patients had decreased patient-reported cough severity as measured by the Cough Visual Analog Scale (VAS) compared to placebo patients, who had increased cough severity at all time points. Did.
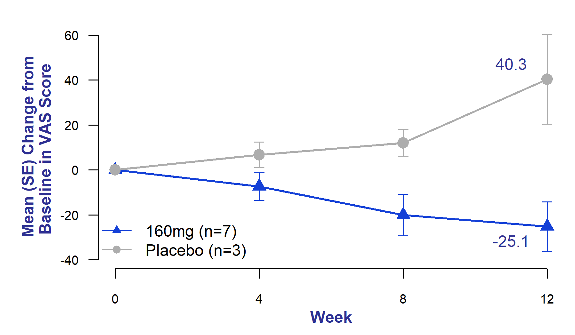

Figure 3. Mean change from baseline in cough severity Visual Analog Scale (VAS)
Bexotegrast 160 mg for 12 weeks
Elevated integrin beta-6 plasma levels are associated with mortality, transplantation, or progression of interstitial lung disease (ILD) defined by a relative decrease of 10% or more in FVC (mL) over a 12-month period.6 PRO-C3, a serum biomarker of type III collagen synthesis, is elevated in IPF patients and is associated with progressive disease.7 At weeks 4 and 12, bexotegrast-treated patients showed reductions in the circulating biomarkers integrin beta-6 and PRO-C3 compared to placebo.
The secondary endpoint of this study was to assess the safety and tolerability of bexotegrast. Bexotegrast at a dose of 160 mg was well tolerated over 12 weeks of treatment and no serious adverse events (SAEs) were reported. The most frequently reported treatment-emergent adverse events (TEAEs) were mild in severity and did not result in study discontinuation.
“These imaging data continue to demonstrate bexotegrast's anti-fibrotic mechanism of action and build on previous results, including our INTEGRIS-IPF Phase 2a study,” said Pliant Therapeutics Chief Medical Officer and CEO of Pliant Therapeutics. said one Dr. Eric Lefebvre.
“The results of this first study using collagen PET imaging to evaluate therapeutic interventions raise the possibility of utilizing this new technology to identify potentially disease-modifying anti-fibrotic IPF therapies in short-term studies. “It highlights the gender,” said Sidney Montesi, M.D., a clinical researcher in the department. Professor of Respiratory and Critical Care Medicine at Massachusetts General Hospital and principal investigator of the trial.
A slide deck containing topline data from this study is available on the “Investors and Media'' section of the Pliant website (www.PliantRX.com).
Phase 2a PET Imaging Study (NCT05621252)
This is a Phase 2a, 12-week, single-center, randomized, double-blind, placebo-controlled study of total collagen deposition levels in the lungs of IPF patients with 160 mg of bexotegrast or placebo once daily. I rated it. . Patients were randomized in a 2:1 ratio (active:placebo) and stratified based on use of standard-of-care IPF therapy. Participants underwent positron emission tomography (PET) imaging using a radioactive tracer. 68Ga-CBP8 binds to total collagen at baseline and week 12. The primary endpoint of the study was to assess baseline changes in type 1 collagen in the lungs, and the secondary endpoints were to assess the safety and tolerability of bexotegrast. The study's exploratory efficacy endpoints evaluated changes in forced vital capacity (FVC) and percent predicted forced vital capacity (FVCpp), changes in patient-reported cough severity, and changes in fibrosis biomarkers. Patients treated with bexotegrast showed improvement across all exploratory efficacy endpoints compared to placebo.
About Pliant Therapeutics
Pliant Therapeutics is a late-stage biopharmaceutical company and a leader in the discovery and development of new therapies for the treatment of fibrotic diseases. Pliant's lead product candidate, bexotegrast (PLN-74809), is an oral small molecule dual-selective inhibitor of αvβ6 and αvβ1 integrins with potential use in the treatment of idiopathic pulmonary fibrosis (IPF) and primary sclerosing cholangitis. It is currently being developed as a major indication. , or PSC. Bexotegrast has received Fast Track and Orphan Drug Designations from the U.S. Food and Drug Administration (FDA) in IPF and PSC, and Orphan Drug Designation from the European Medicines Agency in IPF and PSC. Pliant has initiated his BEACON-IPF, a Phase 2b/3 trial of bexotegrast in IPF. Pl Pliant conducts a Phase 1 trial of his third clinical program, PLN-101095, a small molecule dual selective inhibitor of αvβ8 and αvβ1 integrins being developed for the treatment of solid tumors doing. Pliant has received regulatory approval to conduct a Phase 1 trial of PLN-101325, a monoclonal antibody against integrin α7β1 that targets muscular dystrophy. For more information, please visit www.PliantRx.com. Follow us on social media X, LinkedIn, Facebook, and YouTube.
Forward-looking statements
Statements contained in this press release regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “may,” “will,” “expected,” “anticipated,” “estimate,” “intend,” and similar expressions (as well as future events, conditions, etc.) , or other words or expressions referring to circumstances) are intended to identify forward-looking statements. These statements include those regarding the safety, tolerability, pharmacodynamics, and therapeutic potential of bexotegrast. Future development plans for bexotegrast, PLN-101325 and PLN-101095. The potential for bexotegrast to be a treatment for his IPF and the potential future use of his PET imaging technology to identify IPF treatments. Such statements relate to future events, are based on the Company's current expectations and are therefore subject to various risks and uncertainties and may differ from Pliant Therapeutics' actual results, performance or achievements. may differ materially from what is stated in or implied by this press release. These forward-looking statements relate to the development and commercialization of our product candidates, including delays in our ongoing or planned preclinical or clinical trials, the effects of current macroeconomic and market conditions; may be subject to risks and uncertainties, including: the impact of health epidemics and pandemics, such as COVID-19, on our business, operations, clinical supplies and plans; our dependence on third parties for key aspects of our development operations; risks inherent in the drug development process; risks related to the accuracy of cost estimates and the timing of development, capital requirements and the need for additional financing (including the availability of additional term loans under our financing facilities), and obtaining intellectual property protection for our product candidates; and information about your ability to maintain it. These and other risks are discussed in the sections titled “Risk Factors” and “Management's Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the period ended December 31, 2023. explained. Quarterly Report on Form 10-Q for the period ended March 31, 2024. Each is available on the SEC's website at www.sec.gov. Unless otherwise stated, Pliant is providing this information as of the date of this news release and does not undertake any obligation to update any forward-looking statements contained in this document, whether as a result of new information, future events or otherwise. I am not responsible.
Investor and Media Contact:
Christopher Keenan
Vice President of Investor Relations and Corporate Communications
Pliant Therapeutics, Inc.
ir@pliantrx.com
1 Kuhn C 3rd et al. 1989. Am Rev Respir Dis. Dec;140(6):1693-703.
2 Montesi SB, et al. 2019. Am J Respir Crit Care Med 200:258–261.
3 Juste, A. and others. 2017. Respir Res 18, 74.
Four Van Manen MJG et al. Eur Respir Rev 2016; 25: 278–286.
Five Ryerson CJ et al. Respirology 2011; 16: 969–975.
6 Organ LA et al. Respiratory Resistance 2019 7 12;20(1):148.
7 Bowman WS et al. Lancet Respear Med. 2022 June;10(6):593-602.
The figures accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/aa757c2b-a287-4257-9c63-b9f395966616
https://www.globenewswire.com/NewsRoom/AttachmentNg/6b988e76-1c51-4f89-a68d-51e320a285a2
https://www.globenewswire.com/NewsRoom/AttachmentNg/af7a4480-a23e-4062-85ab-930dac489703




